.jpg)
March 30, 2026
From strain selection to final formulation, industrial probiotic production relies on a precise process to ensure viability, stability, and efficacy.
Probiotics are live microorganisms that provide health benefits when consumed in adequate amounts. Their production is based on an industrial fermentation process that requires strict control of biological and physicochemical parameters to ensure strain viability and effectiveness. This industry, built on advanced technologies, is subject to stringent quality standards to guarantee the purity, stability, and safety of final products.
Probiotics are now essential in several fields:
Their effectiveness relies on rigorous bacterial strain selection and a well-controlled manufacturing process, ensuring their survival and activity after ingestion.
This article provides a detailed overview of the key stages of probiotic manufacturing, emerging trends, and the challenges faced by the industry.
Industrial probiotic production through fermentation is based on a multi-step process aimed at cultivating bacteria under optimal conditions while preserving their functional integrity.
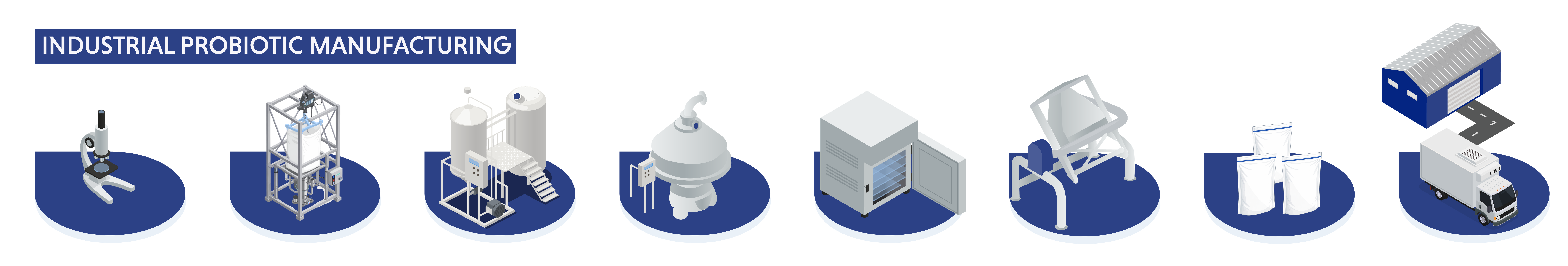
Bacterial strains are selected based on their beneficial properties and their ability to withstand industrial and digestive conditions. They must be stored securely. Several pure strain banks are systematically established to ensure production continuity.
At least three collections are maintained: Master Cell Bank, Working Cell Bank, and Back-Up Cell Bank. These are stored at -80°C in dedicated strain storage freezers located in different secure sites to prevent risks related to power outages or technical failures.

The preparation of the culture medium is a fundamental step that directly impacts fermentation quality and yield. Each bacterial strain requires a specifically formulated nutrient medium composed of carbon sources (carbohydrates), nitrogen sources (peptones, yeast extracts), mineral salts, and essential growth factors.
This medium is prepared in a dedicated area following strict protocols: precise dosing and dissolution of components, pH adjustment, and sterilization (by heat or sterile filtration for heat-sensitive components) to eliminate any microbial contamination.
The exact formulations are typically proprietary to each manufacturer and represent key industrial know-how developed through years of R&D optimization. They are designed to promote both bacterial growth and the expression of functional properties, while meeting regulatory requirements and economic constraints of large-scale production.

The fermentation phase is the core of the process. It involves cultivating bacteria in a strain-specific nutrient medium under controlled conditions:
Industrial fermenters enable batch, fed-batch, or continuous culture, depending on the bacterial species and production objectives.

Bacterial cells are concentrated through centrifugation or tangential filtration. The goal is to remove as much water as possible to facilitate the drying step.
Cryoprotectants and stabilizers are added to the concentrated wet biomass to improve cell resistance to drying and storage conditions.

Probiotics undergo strict quality control, including microbiological and physicochemical analyses.
Different probiotic strains can be blended, depending on the desired formulations, with other ingredients (prebiotics, vitamins, minerals, etc.).
This mixing step ensures a homogeneous distribution of strains, enables the creation of specific combinations for synergistic effects, and prepares the probiotics for incorporation into various dosage forms.

Stabilized probiotics are then offered in various dosage forms, adapted to their final applications:
They can also be directly incorporated into food matrices (yogurts and fermented milk, beverages, nutritional bars, etc.).
Packaging must protect the strains from environmental factors (moisture, oxygen, light) to ensure their stability until consumption.

The supply chain plays a crucial role in preserving probiotic efficacy: products must be transported and stored under controlled temperature and humidity conditions to maintain strain viability until they reach the end consumer.
Depending on dosage forms and target markets, probiotics are distributed through various channels, including retail, pharmacies and parapharmacies, online sales networks, and professional channels serving the food industry and pharmaceutical laboratories.
Shelf life is a key parameter defined upstream by R&D teams, ensuring that a sufficient number of live bacteria remains present until the product’s expiration date—an essential condition for its effectiveness.

Industrial probiotic manufacturing relies on a complex and highly controlled process, from bacterial strain fermentation to stabilization and formulation. The integration of advanced technologies helps improve both efficiency and viability.
By staying at the forefront of innovation and adhering to rigorous quality standards, the probiotic industry can continue to address the challenges of future nutrition and health.
Interested by our services?